High degree-of-freedom system-bath model (a prelude)
Introduction
Locating the Dividing Surface (DS) in chemical reactions has been the focus of many rate constant calculations both for reactions in solution and in vacuum thanks to the Transition State Theory; a classical theory developed by Wigner, Eyring, Evans and Polanyi;[1], [2], [3], [4] that calculates the rate of the reaction as the equilibrium flux of reactive trajectories through that DS.
However, the construction of the DS for 2 DoF systems using the Lyapunov family of unstable POs was presented in a series of papers by Pollak, Pechukas, and Child in the late 1970's to early 1980's.[5], [6], [7], [8], [9] The resulting Periodic Orbit Dividing Surface (PODS) is a hypersurface in phase space arising from the unstable PO. It has been shown to have the required no-recrossing properties described above. Of particular interest was the recognition that as the total energy of trajectories increased, the location of the PODS would change, and that, in general, its projection onto the PES did not need to pass through the index one saddle point that is considered to be the location of the Transition Structure in many chemical models. In the present chapter, we emphasize that it is not only energy which can cause the PODS to move, but at least in some models, its location may also be mass dependent. This feature is of particular relevance to the solvent-derived inertial barrier mentioned above.
(WRITE THE OBJECTIVE OT THE CHAPTER)
Development of the Problem
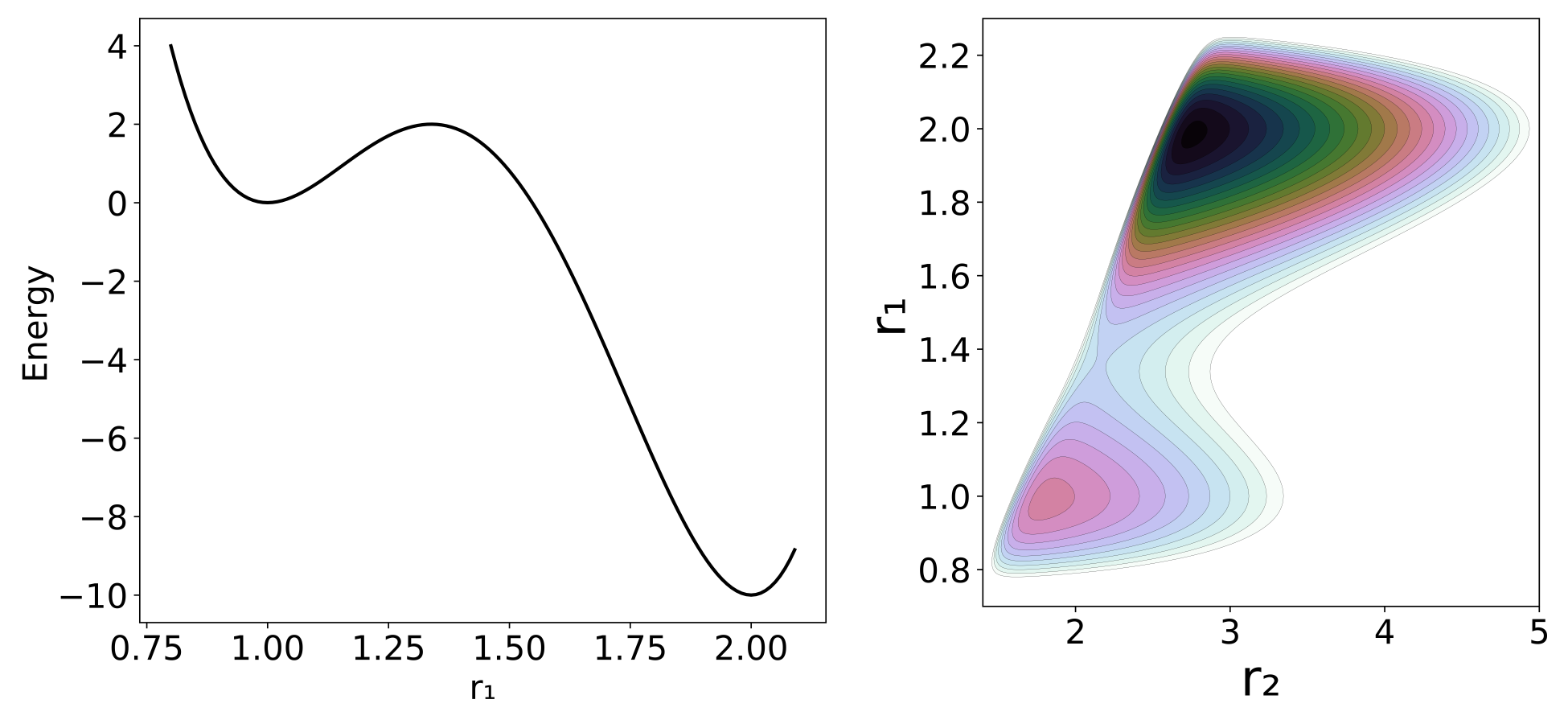
Our model potential (see fig:1) has been introduced in act 2 and consists of a one-dimensional double well oscillator that represents the reactive system, coupled to a one-dimensional harmonic oscillator representing the bath. To facilitat the reading we will reintroduce the details of the model here.

The Hamiltonian that describes the system is as follows:
H(x)=H(r,p)=p212μ1+p222μ2+5∑j=1cjrj−11+c6(c7−r2)2+c8(r2−r1)12where the subscripts 1 and 2 refer to the reactive system and bath oscillators respectively; r=(r1,r2) is the position of the two oscillators and p=(p1,p2) represents the conjugate momenta. The reduced mass of each oscillator is μ, and c are coefficients whose values are listed in fig:1. The potential energy can be divided between V1=∑5j=1cjrj−11 as the potential of the reactive system, V2=c6(c7−r2)2 as the potential of the bath and Vint=c8/(r2−r1)−12 as the interaction between the two; hence V=V1+V2+Vint. The potential of the reactant, shown in fig:2, is chosen to have a minimum at r1=1.0 and a second one at r1=2.0 , with respective potential energies V1=0.0 and V1=−10. The maximum energy is at r1=1.33867 and V1=2.0. The full potential, shown in fig:2, has a saddle point at r1=1.36561 and r2=2.161769 at V=3.47291. The "reactant" minimum occurs at r1=0.98779, r2=1.80661, V=0.77040. The "product" minimum occurs at r1=1.98517, r2=2.75642, V=−6.66284.
Revealing Phase Space Structures
Method 1 - Spatial Lagrangian Descriptors based on Action Integrals
Lagrangian descriptors are calculated on a chosen phase space grid of initial conditions x0 at time t=t0, evolving the trajectories for a fixed forward and backward integration time τ. The general expression of LDs is:
M(x0,t0,τ)=∫t+τt−τF(x(t);x0)dtwhere F(x(t);x0) is a positive and bounded scalar representing a geometrical or physical property of a trajectory with initial conditions x0 and initial time t0; that is integrated over the time interval [t0−τ,t0+τ]. Because we are interested in the DS, and we know that the action is a minimum on its vicinity we will be using an action-like value of the form:
F(x(t);x0)=N∑i=1(pidxidt)1/2where N is the number of DoF, pi and xi are respectively the momenta and position of the DoF i. It is interesting to note that, by finding the modulus of each term separately, we could examine their effect on the LD independently, although that issue is not explored in the present paper. In this case the LD will be:
M(x0,t0,τ)=∫t+τt−τN∑i=1(pidxidt)1/2=∫t+τt−τN∑i=1(v2imi)1/2dtThe application of LD to higher DoF is already being studied in depth.[10], [11] However, these studies revolve around systems with a known Hamiltonian, which is not the case for many chemical processes. Also, the control of every DoF to create the required initial conditions becomes unbearable as soon as you convert the system into a full atomistic model. Therefore, if we want to calculate the LD for complex chemical systems we need a different approach that is described in the following section. As in our previous study the mass of the reactive system (μ1) is set to a value of 1 and the reduced mass of the bath (μ2) will be given values of 0.1, 1 or 10. But in this case we will be using the LD methodology to locate the DS and the τ value will be 1 for μ2=0.1,1 and 2 for μ2=10.
Method 2 - Planar Lagrangian Descriptors based on Action Integrals
The main idea of this method comes from a solution that has been applied in almost every studied chemical reaction. This is, the reduction of the system to a 1 or 2 DoF problem using a Reaction Coordinate (RC). However, as in those chemical studies, the equations of motion are not reduced to a single coordinate but instead the RC is used as a measure of the location of the system at each timestep.
The method was developed with the following assumptions about the problem to be studied:
- The system's Hamiltonian is unknown.
- The system is represented as an atomistic model with Cartesian coordinates.
- We can combine the Cartesian coordinates into one or two collective variables that accurately represent the process we want to model.
Also, the following assumptions are not required but will make the computations much easier:
- The system will have a saddle point from which we will have a rough estimate of where is it.
- The DS is relatively close to that saddle point.
- It is a closed system.
As mentioned previously the definition of the initial conditions of the system can be a major problem when dealing with atoms that have three Cartesian coordinates and velocities each. However, as we want to focus our study in the reactivity of the system, the trajectories in which we are interested have something in common. They all cross a surface orthogonal to the IRC[ref] at the saddle point in their journey. So starting at that point is much easier to assign different initial velocities as a way of exploring part of the phase space.
This part of phase space, is what we will call the reactive phase space which, and by exploring it we can discriminate those phase space structures that have no direct influence in the reactive process.
The general description of the methodology is the following:
- We define 1 or 2 RC that will measure the evolution of the system.
- We establish a set of initial conditions for our RC in the configurational space as our initial point.
- From this point, we integrate several trajectories with different momenta of a time length longer than τ, both forward and backward in time.
- For each trajectory of length T we can calculate the LD with a value of τ for at least a number (T−2τ)/dt of initial conditions that have enough trajectory length before and after them.
The amount of trajectories and their length will strongly depend on the problem and the time it takes to the trajectory to explore the reactive phase space. But, in principle will follow the rule of the longer the better.
In this method, the evolution of the trajectory defines the initial conditions and not the user. Thus, avoiding the problem of defining many initial conditions. Also, although here we talk about having a single point as initial condition there is no reason for not including a second or several points in configurational space as initial conditions. This will be most useful when the DS is far from our initial condition as we will see in the System bath model.
Implications for Reaction Dynamics
Method 1
The images shown in fig:3 where obtained by the above mentioned grid methodology. In those figures, for each point in the plot, the momenta of the bath DoF was equal to 0. The location of the saddle point (blue line) and the PO associated to it (red line) were included in the plots to better understanding of the information obtained. Of significant relevance are the plots obtained for the position and momenta of the reactive DoF (a plots in fig:3). In those plots we can see how the invariant manifolds converge at the PO that encloses the DS forming a crossing point that indicates its location.
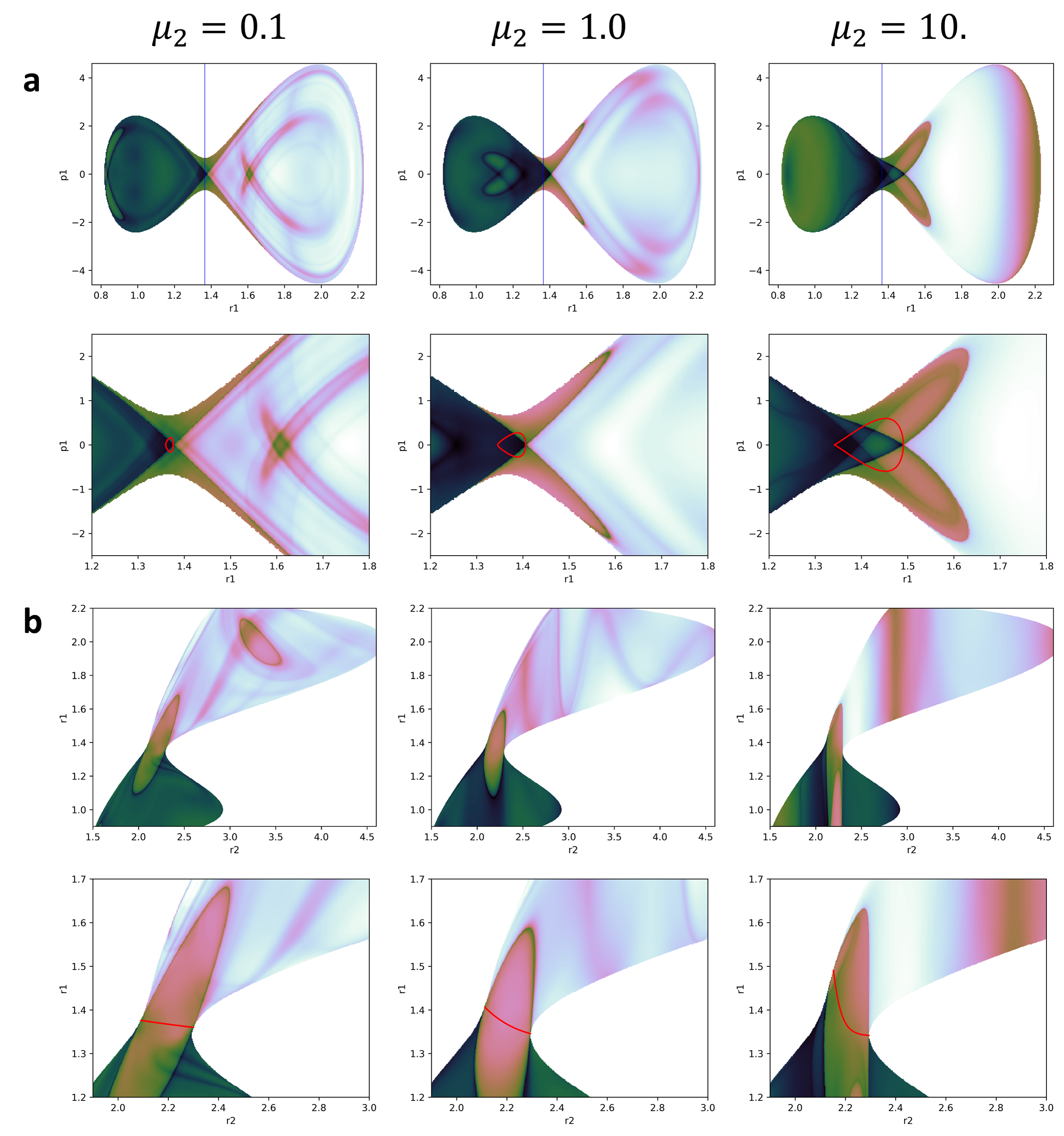
It is important to note that this plots represent a section of the full phase space in which all the kinetic energy is in the reactive DoF. Thus, all we can expect to see from the PODS is the intersection with this section. Of course this is not a big problem, as it is very easy to change the value of the momenta of the bath at which the LD is calculated.
Method 2
We could suppose that the 2DoF from the described system are instead 2 collective coordinates obtained from a system of multiples DoF. We then proceed to calculate the LD as described in our second method, first to check if we could replicate the results from the previous method and then to analyze the extra information we are generating with it.
In fig:4 we can see how the plots are very similar to those obtained in fig:3. The main difference appears as we increase the mass of the bath where this effect reduces the ergodicity of the system. Even in those cases, there are similarities between plots in the places visited by the trajectories.
 obtained with the second methodology](figures/LDSecondSection.png)
But this is just a small amount of information that we have obtained from the calculations. The full set of values that this method produces can be projected into a single plot. Obviously these projections have so many dynamical structures depicted in them that it is very difficult to see anything. But with a bit of cleaning we can obtain a nice picture of the dynamical structure of the system. The plots shown in fig:5 where obtained by projecting in configurational space all the values obtained from the calculations. However, for each value proyected in the same point only the minimum value was represented. This is because the value of the LD is expected to be a minimum in the vicinity of the PODS. These plots show a clear picture of the behaviour of the trajectories after crossing the DS but also they have a clear definition of the projection of the PODS in configurational space.
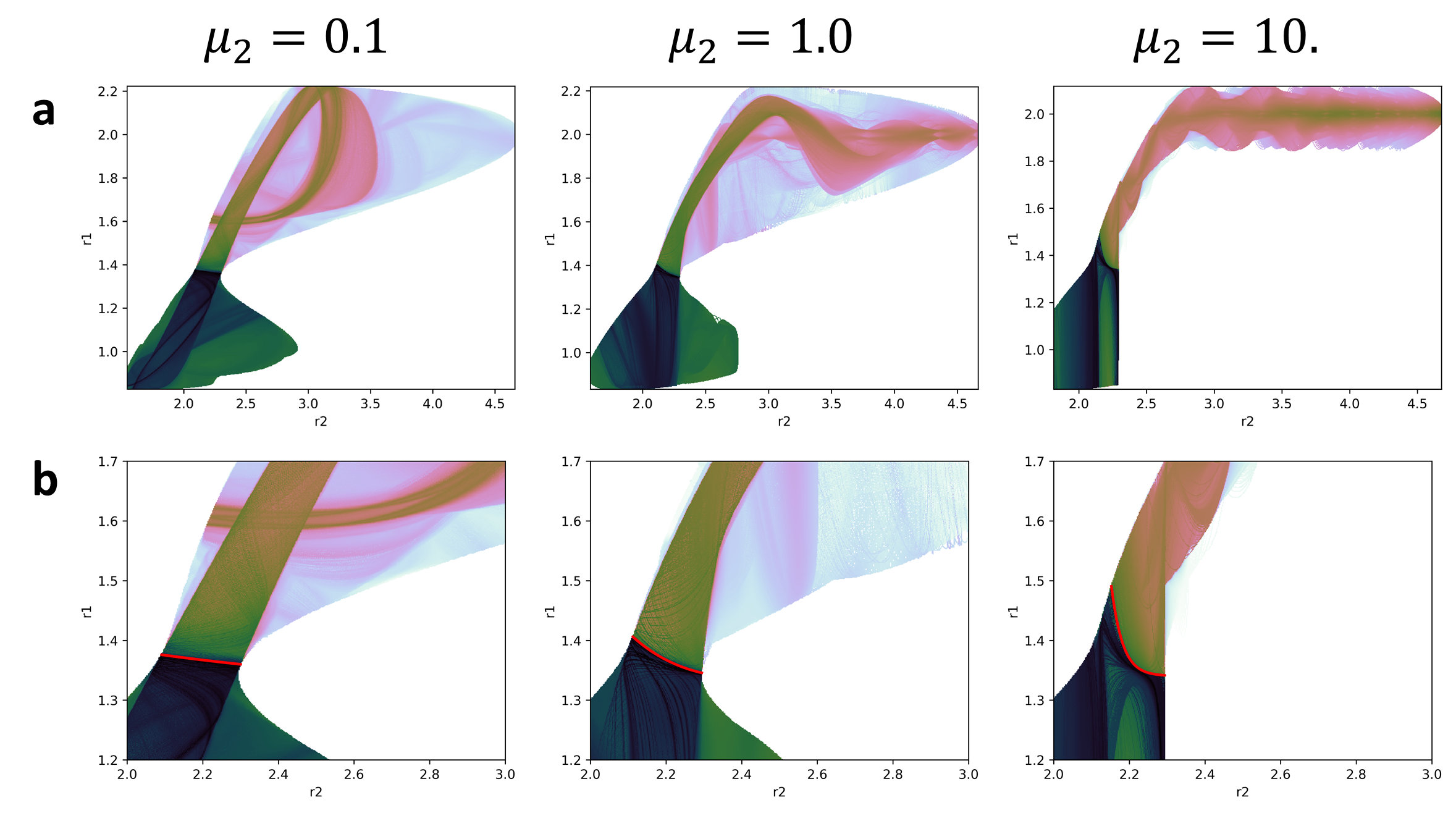
Moreover, if take a slice of the plots in fig:5 we can visualize the effect of the DS in the value of the LD (see fig:6). This is quite obvious in fig:5 but it will be very useful in multiples degrees of freedom where there will be much more noise.
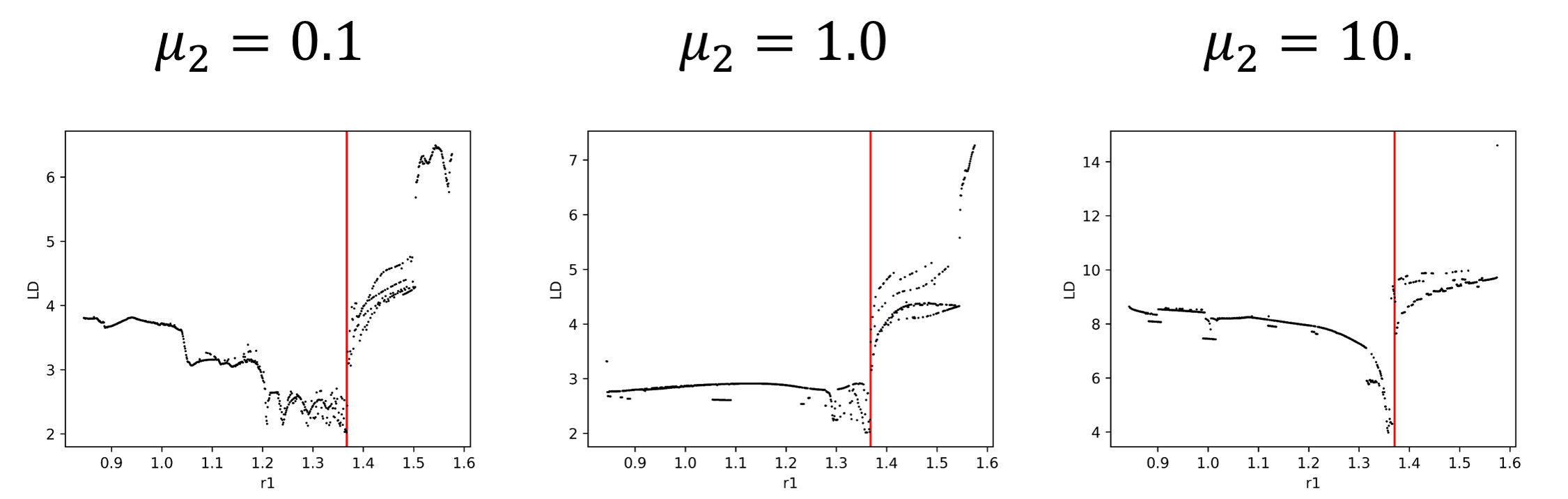
We have shown that the DS can be easily identified by the calculation of LD, not only with the grid methodology, but also with our version of the method. It is expected that for multidimensional problems the results will not be as clear and sharp as with this simple model. After all, we are including a significant amount of DoF that will introduce a lot of noise in the calculations. However, if we can identify the DS signal in the LD calculation, then we can begin to understand the dynamical properties that make the system behave as it does, like in the former case, the coupling between the bath and the reactive system.
References
- H. Pelzer and E. Wigner, “The speed constansts of the exchange reactions,” Z. Phys. Chem. B., vol. 15, p. 445, 1932.
- H. Eyring, “The Activated Complex in Chemical Reactions,” J. Chem. Phys., vol. 3, no. 2, pp. 107–115, 1935.
- M. G. Evans and M. Polanyi, “Some applications of the transition state method to the calculation of reaction velocities, especially in solution,” Trans. Far. Soc., vol. 31, no. 0, pp. 875–894, 1935.
- S. Glasstone, K. J. Laidler, and H. Eyring, The theory of rate processes: the kinetics of chemical reactions, viscosity, diffusion and electrochemical phenomena. New York: McGraw-Hill Book Company, inc., 1941.
- P. Pechukas and E. Pollak, “Trapped trajectories at the boundary of reactivity bands in molecular collisions,” J. Chem. Phys., vol. 67, no. 12, pp. 5976–5977, 1977.
- E. Pollak and P. Pechukas, “Transition states, trapped trajectories, and classical bound states embedded in the continuum,” J. Chem. Phys., vol. 69, no. 3, pp. 1218–1226, 1978.
- P. Pechukas and E. Pollak, “Classical transition state theory is exact if the transition state is unique,” J. Chem. Phys., vol. 71, no. 5, pp. 2062–2068, 1979.
- E. Pollak, M. S. Child, and P. Pechukas, “Classical transition state theory: A lower bound to the reaction probability,” J. Chem. Phys., vol. 72, no. 3, pp. 1669–1678, 1980.
- P. Pechukas, “Recent Developments in Transition State Theory,” Ber. Bunsen-Ges. Phys. Chem., vol. 86, no. 5, pp. 372–378, 1982.
- S. Naik and S. Wiggins, “Finding normally hyperbolic invariant manifolds in two and three degrees of freedom with Hénon-Heiles-type potential,” Phys. Rev. E, vol. 100, no. 2, p. 022204, 2019.
- S. Naik, V. J. García-Garrido, and S. Wiggins, “Finding NHIM: Identifying High Dimensional Phase Space Structures in Reaction Dynamics using Lagrangian Descriptors.” 2019.
